BETHESDA, Maryland, Dec. 1, 2016 /PRNewswire/ — Cannabics Pharmaceuticals Inc. (CNBX) announced today that results from the company’s Cancer HTS research indicates that specific ratios of Cannabinoids led to Apoptosis in MDA-MB-231 Breast Cancer cell viability.
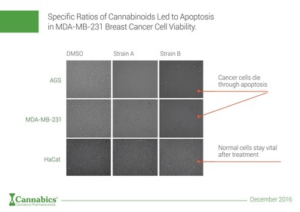
Cannabics Pharmaceuticals is currently developing a personalized treatment based upon screening of patients’ biopsy and delivering specified cannabinoid medicine which contains anti-tumor properties. It is believed this technology will enable oncologist and other treating physicians to provide cancer patients with a personalized anti-cancer cannabinoid based therapy.
“We are building a data base of patients and compounds, the screening data and the clinical efficacy will be centralized and in proprietary software,” said Itamar Borochov, CEO of Cannabics Pharmaceuticals Inc. “Results from the screening of numerous cancer types will produce new data and new patent applications of compounds with anti-cancer properties.”
About Cannabics Pharmaceuticals Inc.
Cannabics Pharmaceuticals (CNBX), a U.S based public company, is dedicated to the development of Personalized Anti-Cancer and Palliative treatments. The Company’s R&D is based in Israel, where it is licensed by the Ministry of Health for its work in both scientific and academic research as well as current clinical studies underway at Rambam Hospital. The Company’s scientific focus is on identifying and harnessing the therapeutic properties of specific natural Cannabinoids. Cannabics’ vision is to create individually tailored therapies for cancer patients, utilizing advanced screening systems and personalized bioinformatics tools.
Disclaimer:
Certain statements contained in this release may constitute forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Such statements include, but are not limited to statements identified by words such as “believes,” “expects,” “anticipates,” “estimates,” “intends,” “plans,” “targets,” “projects” and similar expressions. The statements in this release are based upon the current beliefs and expectations of our company’s management and are subject to significant risks and uncertainties. Actual results may differ from those set forth in the forward-looking statements. Numerous factors could cause or contribute to such differences, including, but not limited to, results of clinical trials and/or other studies, the challenges inherent in new product development initiatives, the effect of any competitive products, our ability to license and protect our intellectual property, our ability to raise additional capital in the future that is necessary to maintain our business, changes in government policy and/or regulation, potential litigation by or against us, any governmental review of our products or practices, as well as other risks discussed from time to time in our filings with the Securities and Exchange Commission, including, without limitation, our latest 10-Q Report filed on July 13th, 2016. We undertake no duty to update any forward-looking statement or any information contained in this press release or in other public disclosures at any time.
