TEL AVIV, Israel and BETHESDA, Maryland, March 2, 2020 /PRNewswire/ — Cannabics Pharmaceuticals Inc. (OTCQB: CNBX), a leader in personalized cannabinoid medicine focused on cancer and its side effects, released an update today regarding its pre-clinical studies held at the company’s High Throughput Screening (HTS) facility in Israel.
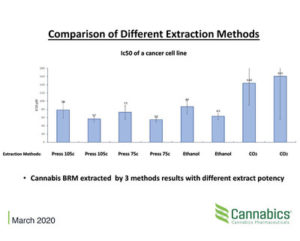
The preliminary results indicate that different extraction methods result in varied antitumor properties of cannabis BRM (Botanical Raw Material) exhibited in necrotic effects on gastrointestinal cancer cells.
Edible products in the cannabis industry are infused with cannabis resin extracted by three main methods: CO2, ethanol and pressure. These methods have advantages and disadvantages however their effect on the medicinal properties of cannabis for cancer patients were never clinically tested.
The current study indicated that heat and pressure extracts a more potent fraction of the plant than CO2 and ethanol in most of the cannabis strains tested. The study will further examine the chemical bases for this differentiation while its implications are clear. The development of medicine based on botanical material is confound to various parameters such as genetics, growth conditions and according to this study – extraction methods. The company also examines the effects of post extraction manipulation such as decarboxylation, winterization and formulation on the invitro toxic effects.
About Cannabics Pharmaceuticals
Cannabics Pharmaceuticals Inc. (OTCQB: CNBX) is a U.S public company that is developing a platform that leverages novel drug-screening tools to create cannabinoid-based therapies for cancer that are more precise to a patient’s profile. By developing tools to assess effectiveness on a personalized basis, Cannabics is helping to move cannabinoids into the future of cancer therapy. The Company’s R&D is based in Israel, where it is licensed by the Ministry of Health to conduct scientific and clinical research on cannabinoid formulations and cancer.
For the latest updates on Cannabics Pharmaceuticals follow the Company on Twitter@Cannabics, Facebook@CannabicsPharmaceuticals, LinkedIn, and on Instagram @Cannabics_Pharmaceuticals.
Disclaimer:
Certain statements contained in this release may constitute forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and other U.S. Federal securities laws. Such statements include but are not limited to statements identified by words such as “believes,” “expects,” “anticipates,” “estimates,” “intends,” “plans,” “targets,” “projects” and similar expressions. The statements in this release are based upon the current beliefs and expectations of our Company’s management and are subject to significant risks and uncertainties. Actual results may differ from those outlined in the forward-looking statements. Numerous factors could cause or contribute to such differences, including, but not limited to, results of clinical trials and other studies, the challenges inherent in new product development initiatives, the effect of any competitive products, our ability to license and protect our intellectual property, our ability to raise additional capital in the future that is necessary to maintain our business, changes in government policy and regulation, potential litigation by or against us, any governmental review of our products or practices, as well as other risks discussed from time to time in our filings with the Securities and Exchange Commission including, without limitation, our latest 10-Q Report filed January 14th, 2020. We undertake no duty to update any forward-looking statement or any information contained in this press release or other public disclosures at any time. Finally, the investing public is reminded that the only announcements or information about Cannabics Pharmaceuticals Inc., which are condoned by the Company, must emanate from the Company itself and bear our name as its source.
